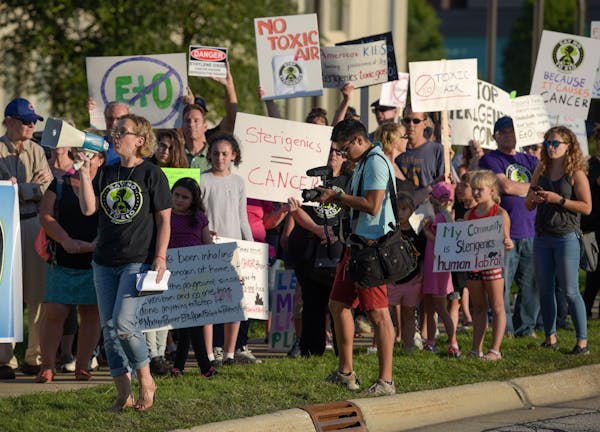Half of all medical devices used in the United States — from bandages to implantable computers — are sterilized with a toxic gas that has come under scrutiny over concerns about air pollution that could cause cancer.
Now, Minnesota Attorney General Keith Ellison has joined a coalition of 11 attorneys general asking the federal Environmental Protection Agency to crack down on emissions of the chemical, ethylene oxide (abbreviated as EO or EtO), which is used on an industrial scale to kill pathogens on medical devices, food spices and other products.
"Fortunately, Minnesota doesn't rank high on EtO emissions, but science is pointing to it being a far more potent carcinogen than previously thought, which puts people at greater risk than previously thought," Ellison said in an e-mailed statement. "The fact that the control equipment that federal regulations currently mandate isn't containing EtO emissions to safe levels is hurting not only local communities, but also Minnesota's medical-device industry."
Med-tech trade groups have warned in stark terms that creating additional controls on EtO or abruptly cutting back on its use could trigger widespread shortages of lifesaving medical devices.
"If someone needs a pacemaker, and they can't get one, that creates its own public health crisis," said Shaye Mandle, CEO of the Medical Alley Association trade group in Golden Valley. "We are not saying, don't do anything on EtO. We are saying, if you eliminate it tomorrow in the absence of an alternative — and today there is no alternative — then medical devices that use this sterilization methodology … wouldn't be able to go to market."
Other sterilization methods like heat and radiation can damage the soft plastics and electronics in medical devices. The EtO industry said the gas is uniquely useful because it's highly effective at killing pathogens and can penetrate special packaging to sterilize the insides of devices and surgical kits. The gas enables manufacturers to meet the international "sterility assurance" benchmark of having less than a 1-in-a-million chance of harboring a viable pathogen.
The Food and Drug Administration is already working with companies to accelerate development and review of 11 new sterilization ideas for the medical device industry, but the federal EPA regulates EtO emissions. The EPA is now analyzing whether to lower EtO emission limits and tighten rules on how the chemical is handled.
EtO represents a conundrum for industry. It's widely believed to be a potent carcinogen for workers who handle it and people who live downwind of EtO plants, yet it's used to sterilize more than 20 billion health care products a year, preventing the spread of infectious germs.
"For many devices … EO is the only method that has been shown to provide effective sterilization without causing damage to the device," Medtronic environmental health and safety director Peter J. Jansen said in a Feb. 10 comment letter to the EPA. Medtronic, the world's largest medical device company, has its operational headquarters in Minnesota.
Medtronic was one of nine companies surveyed by the EPA last year about its use of EtO, along with Medline Industries, Becton Dickinson (BD) and Sotera Health, an official with the EPA confirmed. While most med-tech companies use outsourced facilities for EtO sterilization, Medtronic and Boston Scientific also run their own EtO sterilizers, federal records show.
The United States has about three dozen EtO plants, including a Steris AST contract-sterilization facility in Coon Rapids. But the number of device-sterilizer plants currently using EtO nationally has been dropping quickly.
Sotera's Sterigenics plant in Willowbrook, Ill., permanently closed last year following strong community protest and state regulatory actions. Meanwhile, BD's plants in Smyrna and Covington, Ga., have come under fire from state officials and community groups for EtO emissions. Viant Medical agreed to close its EtO plant in Grand Rapids, Mich., earlier this year, while Medline's EtO plant in Waukegan, Ill., is reopening following a new state law that imposed stricter emission standards.
Minnesota AG Ellison has joined Illinois Attorney General Kwame Raoul and nine other state AGs in urging the EPA to essentially adopt Illinois' tougher statute, called the Matt Haller Act, and make it apply nationally. Among other things, the Haller Act requires EtO users in Illinois to capture 100% of EtO emissions from the rooms where it's used, reduce EtO emitted into the atmosphere to 0.2 parts per million, and conduct continuous emissions monitoring and ambient air testing.
"The control measures in [existing federal regulations on EtO] are not sufficiently protective of human health in light of new, compelling information," the attorneys' general letter says.
The compelling information is a December 2016 risk analysis commonly referred to the IRIS assessment, which concluded that EtO was highly likely to be a mutagenic human carcinogen even at low "environmental" levels of exposure, based on a 2004 study of the health of 18,000 people who worked at commercial sterilization plants.
Washington-based med-tech trade group AdvaMed says the IRIS report increased the suspected "cancer potency" of EtO 30-fold. That increase was set using evidence from a single study that documented many thousands of workers who were chronically exposed to "extraordinarily high" concentrations of EtO and yet did not get cancer. Only those exposed to "the very highest concentrations for the very longest periods of time," got cancer, AdvaMed wrote.
Pro-industry trade groups like AdvaMed say regulating EtO use based on the IRIS analysis would go too far, because people living downwind from EtO plants are already exposed to greater concentrations, like the EtO produced by human metabolism and by combustion in the environment.
"EtO is also a normal component of ambient air," AdvaMed wrote to the EPA on Feb. 18. "The 2016 IRIS assessment of EtO should NOT be used for regulatory purposes because it defies science and common sense in the context of everyday human exposures."
One commenter urged EPA to use IRIS risk estimates, noting that the medical device industry's own standards document, ISO 10993-7, says genetic factors may determine susceptibility to enhanced cancer risks from EtO. The comment letter, submitted anonymously to the EPA, said impaired function of an enzyme called GSTT1 in some people leaves them unable to "detoxify" EtO via metabolism, putting them at greater risk of developing health problems from exposure to it.
"No amount of ethylene oxide is safe for the public," the letter concluded.
Joe Carlson • 612-673-4779
Amazon cloud computing unit plans to invest $11 billion to build data center in northern Indiana

Xcel profits after job cuts, but Texas wildfire costs could be 'adverse'
Southwest will limit hiring and drop 4 airports after loss. American Airlines posts 1Q loss as well
That's rich: Rise in all-cash transactions helping drive price gains for most expensive US homes